Theories
There are 3 theories that define acids and bases.

The earliest, being the Arrhenius Theory, introduced in 1887 by Svante Arrhenius.
The Arrhenius Theory states:
The Arrhenius Theory states:
- Acids are substances that produce hydrogen ions (or H+ ions) when in water
- Bases are substances that produce hydroxide ions (or OH- ions) when in water

The second theory is the Brønsted-Lowry theory, which was introduced in 1923.
The Brønsted-Lowry Theory states:
The Brønsted-Lowry Theory states:
- Acids are proton [hydrogen ions (or H+ ions)] donors
- Bases are proton [hydroxide ion (OH-)] receivers

The third theory is the Lewis Theory, which was also introduced in 1923.
The Lewis Theory states:
The Lewis Theory states:
- Acids are electron pair acceptors
- Bases are electron pair donors
These theories serve to define acids and bases. There are many properties of acids and bases which can further
identify substances as acids or bases.
identify substances as acids or bases.
Properties of Acids & Bases
Uses of Acids and Bases:
|
Acids:
Bases:
|
|
Strength vs. Concentration
Strength, refers to an acid or bases ability to dissociate in a solution. A strong acid or base dissociates completely in a solution, and a weak acid or base dissociates only partially, or not at all in a solution.
Concentration, refers to the amount of substance per unit volume. Concentration is often expressed as Molarity.
Molarity is the molar concentration of either H+ ions, or OH- ions, depending on whether it is an acid or a base.
Concentration, refers to the amount of substance per unit volume. Concentration is often expressed as Molarity.
Molarity is the molar concentration of either H+ ions, or OH- ions, depending on whether it is an acid or a base.
Conjugate Acids / Bases
Conjugate acids and bases refer to the substances created in a chemical reaction.
A conjugate acid is the substance created after the base receives an H+ ion in a chemical reaction.
A conjugate base is the substance created after the acid donates an H+ ion in a chemical reaction.
A conjugate acid is the substance created after the base receives an H+ ion in a chemical reaction.
A conjugate base is the substance created after the acid donates an H+ ion in a chemical reaction.
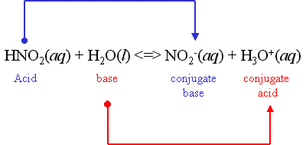
In the equation to the left, you can see and acid (HNO2) reacting with a base (H2O). To identify the conjugate acid and base, look at the right side of the equation.
The acid (HNO2) donates its H+ ion to become the conjugate base (NO2).
The base (H2O), receives an H+ ion to become the conjugate acid (H3O).
The acid (HNO2) donates its H+ ion to become the conjugate base (NO2).
The base (H2O), receives an H+ ion to become the conjugate acid (H3O).
Alkalis
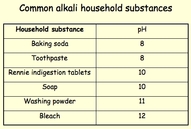
The term alkali refers to a sub-group of bases.
They are bases that dissolve in water.
Alkalinity is a measure of a solutions ability
to neutralize an acid.
They are bases that dissolve in water.
Alkalinity is a measure of a solutions ability
to neutralize an acid.
Indicators
There are several indicators which serve to identify solutions as acidic, basic or neutral.
They are able to indicate these conditions by noticeably changing color.
The 3 main indicators for acids and bases are phenolphthalein, litmus paper and universal indicator.
They are able to indicate these conditions by noticeably changing color.
The 3 main indicators for acids and bases are phenolphthalein, litmus paper and universal indicator.

Phenolphthalein is a liquid indicator which changes the color of the solution it is placed in.
When drops are placed in an acid solution, it remains clear.
When drops are placed in a neutral solution, it remains clear.
When drops are placed in a basic solution, it turns purple.
When drops are placed in an acid solution, it remains clear.
When drops are placed in a neutral solution, it remains clear.
When drops are placed in a basic solution, it turns purple.
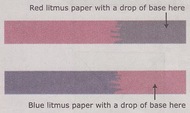
Litmus paper is a paper indicator that is placed in solutions to determine whether they are acidic, basic or neutral. It comes in two colors: red and blue.
Red litmus paper turns blue when it is placed in a base.
Blue litmus paper turns red when it is placed in an acid.
Red litmus paper turns blue when it is placed in a base.
Blue litmus paper turns red when it is placed in an acid.
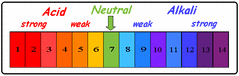
Universal indicator is the third common indicator which identifies whether the solution is acidic, basic or neutral, as well as its pH value.
red - orange = strong acid
orange - yellow = weak acid
green = neutral
light blue - dark blue = weak base (alkali)
dark blue - dark purple = strong base (alkali)
red - orange = strong acid
orange - yellow = weak acid
green = neutral
light blue - dark blue = weak base (alkali)
dark blue - dark purple = strong base (alkali)
Feeling Confident?


